|
Current research continues into the structure of the atom. These subatomic particles cannot be freed and studied in isolation. With a lack of electrical charge, the neutron is not repelled by the cloud of electrons or by the nucleus, making it a useful tool for probing the structure of the atom.Įven the individual protons and neutrons have internal structure, called quarks. The neutron carries no electrical charge and has the same mass as the proton. It was discovered by a British physicist, Sir James Chadwick. Protons, neutrons, and electrons are subatomic particles, as are all hadrons and. The neutron is the other type of particle found in the nucleus. Any of various particles of matter that are smaller than a hydrogen atom. A proton has a mass of 1.67 × 10 -27 kilograms. The number of protons in the nucleus of an atom determines what kind of chemical element it is. The proton's charge is equal but opposite to the negative charge of the electron. The smallest part of an element that can exist alone Proton. The existence of a positively charged particle, a proton, in the nucleus was proved by Sir Ernest Rutherford in 1919. in nuclear and particle physics is the barn (or b) defined as. One of the two types of particles found in the nucleus is the proton. Subatomic physics is the part of this story starting with nuclei and continuing on to the. The mass of an electron is 9.1 × 10 -31 kilograms. Most properties of atoms are based on the number and arrangement of their electrons. The negatively charged electrons follow a random pattern within defined energy shells around the nucleus. Thomson discovered the existence of the electron, marking the beginning of modern atomic physics. The diameter of an atom is approximately 2-3 Å. 
A convenient unit of length for measuring atomic sizes is the angstrom (Å), which is defined as 1 × 10 -10 meters. Macroscopic particles are particles that are larger than atoms or. There are many different types of particles, with different particle sizes and properties. In particle physics, an elementary particle is a particle which cannot be split up into smaller pieces. This nucleus contains most of the atom's mass and is composed of protons and neutrons (except for common hydrogen which has only one proton). A particle is a tiny bit of matter that makes up everything in the universe. Electrons also play an important role in how atoms bond to each other to form molecules and will therefore be discussed in more detail in future sections of this module.An atom is a complex arrangement of negatively charged electrons arranged in defined shells about a positively charged nucleus. In general, atoms like to have the same number of electrons as they have protons in order to be electrically balanced. The matter is made up of inconceivably small atoms, and yet scientists found atoms contain even smaller subatomic particles, including electrons, protons. Electrons are negatively charged subatomic particles that are as negative as protons are positive. In the graphic below you will notice that each of the three elements have different numbers of protons (red spheres) and neutrons (blue spheres).ÂĪs shown in the figure above, electrons surround the nucleus. If all protons are identical and all neutrons are identical, then what makes the atoms of two different elements different from each other? For example, what makes a hydrogen atom different from a helium atom? As mentioned in our discussion of elements and the periodic table, the number of protons and neutrons in the nucleus give the atoms their specific characteristics. Neutrons have no electrical charge and are said to help hold the protons together since protons are positively charged particles and should repel each other. Protons have a positive electrical charge, so they are often represented with the mark of a "+" sign. All protons are identical to each other, and all neutrons are identical to each other. 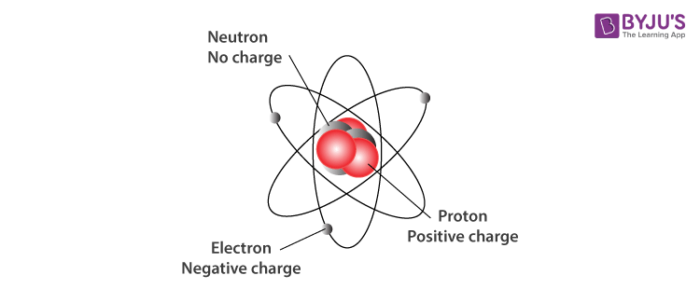
Protons and neutrons make up the nucleus of an atom. 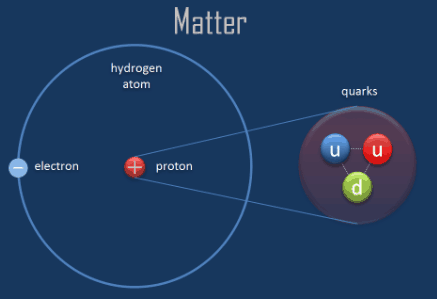
The center of the atom is called the nucleus. Protons and Neutrons The three main subatomic particles that form an atom are protons, neutrons, and electrons. There are three subatomic particles: protons, neutrons and. Particles that are smaller than the atom are called subatomic particles. Given that these particles make up atoms, they are often referred to as subatomic particles. Now that we have talked the basics of atoms and elements, let's talk about the particles that make up the atom. Typically, an atom can be broken down into three subatomic particles, namely: protons, electrons, and neutrons. Discuss the positions of these particles within the atom and what electric charge they carry, if any. Subatomic Particles - Definition, Discovery, and Key Features Chemistry Structure of Atom Subatomic Particles Subatomic Particles A Subatomic particle is nothing but a particle which is smaller than an atom in size.List the three main subatomic particles of an atom.After reading this section you will be able to do the following:
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
